31 July 2015
Abstract
Born et al. [1] intranasally administered three peptides, MSH/ACTH, vasopressin, and insulin, and demonstrated that intranasal administration allowed these peptides to pass into the cerebrospinal fluid (CSF). Neither MSH/ACTH or insulin passed into the blood, and plasma glucose did not change after insulin administration. Born et al. [1] used the linear trapezoidal method, a non-compartmental method, to estimate bioavailability (area under the curve, AUC) of the peptides in the CSF and blood. The vasopressin and insulin data did not admit of another method, compartmental analysis, for assessing kinetics. However, the MSH/ACTH data were suitable for compartmental analysis. We used Born’s published data and found that the half life of MSH/ACTH in the nasopharynx (t1/2 ka) was 3.15 min. The half life in the brain (t1/2 alpha) was 97.4 minutes, and the half life in the CSF (t1/2 beta) was 475 minutes. Because the molecular weights of MSH/ACTH (4540 daltons) and insulin (5800 daltons) are similar and both are polypeptides, we expect MSH/ACTH and insulin to have similar kinetics when administered intranasally. Intranasal insulin improves cognitive function in both normal subjects and Alzheimer’s disease patients, yet little is known regarding the central nervous system kinetics of intranasal insulin. Our results derived from MSH/ACTH may inform further studies.
Born et al. [1] intranasally administered three peptides, MSH/ACTH, vasopressin, and insulin, and demonstrated that intranasal administration allowed these peptides to pass into the cerebrospinal fluid (CSF). Born et al. [1] used the linear trapezoidal method, a non-compartmental method, to estimate bioavailability (area under the curve, AUC) of the peptides in the CSF and blood. The vasopressin and insulin data did not admit of another method, compartmental analysis, for assessing kinetics. However, the MSH/ACTH data were suitable for compartmental analysis. We took the seven data points from the 10 mg MSH/ACTH curve in Figure 1a of Born et al. [1] and performed a two compartment (brain, CSF) extravascular infusion zero order absorption analysis [2]. The two compartment model can be applied as a reasonable approximation of intranasal peptide delivery, as is demonstrated in Figure 1.
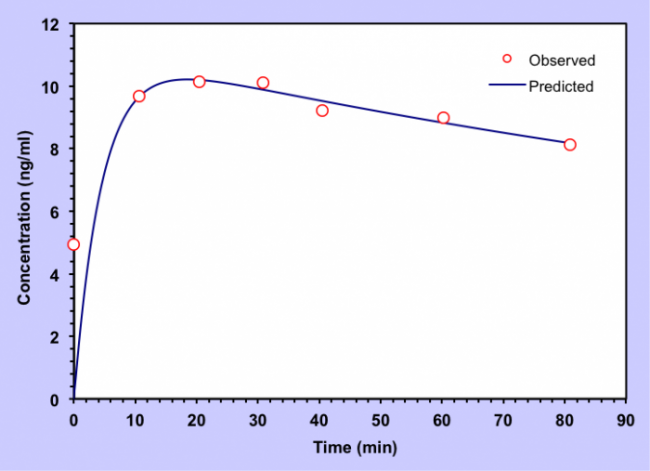
Figure 1. MSH/ACTH accumulation in CSF after a 10 mg intranasal infusion, 7 observed data points from Born et al. Note that the predicted curve from the two compartment extravascular infusion zero order absorption analysis is a good fit to the actual data points.
We performed the analysis with pK solver, an add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel [3]. We found that the half life of MSH/ACTH in the nasopharynx (t1/2 ka) was 3.15 min. The half life in the brain (t1/2 alpha) was 97.4 minutes, and the half life in the CSF (t1/2 beta) was 475 minutes. MSH/ACTH is a peptide composed of 39 amino acids. Insulin is composed of two polypeptide chains, subunits A and B. Subunit A consists of 21 amino acids, whereas subunit B consists of 30 amino acids. The chains are connected by two disulfide bridges. Because the molecular weights of MSH/ACTH (4540 daltons) and insulin (5800 daltons) are similar and both are polypeptides, we expect MSH/ACTH and insulin to have similar kinetics when administered intranasally. Insulin signaling is impaired in Alzheimer’s disease [4]. In 1996, Craft et al. showed that intravenous insulin improved cognition [5]. Moreover, intranasal insulin improves cognitive function in both normal subjects and Alzheimer’s disease patients without the hypoglycemia associated with intravenous insulin [6, 7]. Yet little is known regarding the central nervous system kinetics of intranasal insulin. Our results derived from MSH/ACTH may inform further studies.
Steven Lehrer, MD
Fermata Pharma, Inc.
Peter Rheinstein, MD, MS, JD
Severn Health Solutions
Correspondence to: Dr. Steven Lehrer, Fermata Pharma, Inc, 30 West 60th Street, New York, New York 10023-7909 or steven@fermatapharma.com
Disclosure: Dr. Lehrer has filed a patent application on the use of preservative-free nasal insulin for the treatment of Alzheimer’s disease.
References
[1] Born J, Lange T, Kern W, McGregor GP, Bickel U, Fehm HL (2002) Sniffing neuropeptides: a transnasal approach to the human brain. Nat Neurosci 5, 514-516.
[2] Hacker M, Messer II WS, Bachmann KA (2009) Pharmacology: principles and practice, 1st ed. Academic Press, Burlington, MA.
[3] Zhang Y, Huo M, Zhou J, Xie S (2010) PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comput Methods Programs Biomed 99, 306-314.
[4] Candeias E, Duarte AI, Carvalho C, Correia SC, Cardoso S, Santos RX, Placido AI, Perry G, Moreira PI (2012) The impairment of insulin signaling in Alzheimer's disease. IUBMB Life 64, 951-957.
[5] Craft S, Newcomer J, Kanne S, Dagogo-Jack S, Cryer P, Sheline Y, Luby J, Dagogo-Jack A, Alderson A (1996) Memory improvement following induced hyperinsulinemia in Alzheimer's disease. Neurobiol Aging 17, 123-130.
[6] Benedict C, Hallschmid M, Schmitz K, Schultes B, Ratter F, Fehm HL, Born J, Kern W (2006) Intranasal Insulin Improves Memory in Humans: Superiority of Insulin Aspart. Neuropsychopharmacology 32, 239-243.
[7] Claxton A, Baker LD, Hanson A, Trittschuh EH, Cholerton B, Morgan A, Callaghan M, Arbuckle M, Behl C, Craft S (2015) Long-acting intranasal insulin detemir improves cognition for adults with mild cognitive impairment or early-stage Alzheimer's disease dementia. J Alzheimers Dis 44, 897-906.






